Molar Mass Of Ni Dmg 2 Physical Chemistry Virtual Lab 
A sintered glass crucible of mass 14.1564 g. Enter your answer to 2 decimal placesA solid sample of a complex salt, with ideal composition Ni(NH3)6(NO3)2, was analyzed for nickel by dissolution in slightly acidic solution and precipitation as the dimethylglyoximate, Ni(DMG)2. Quantatitative determination of nickel in a compound by gravimetric analysis 1 answer below » A 10.00 ml sample of an unknown aqueous nickel (II) solution was treated and found to produce 0.0532 g of Ni(DMG)2(Molar mass=288.9155g/mol) precipitate. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. 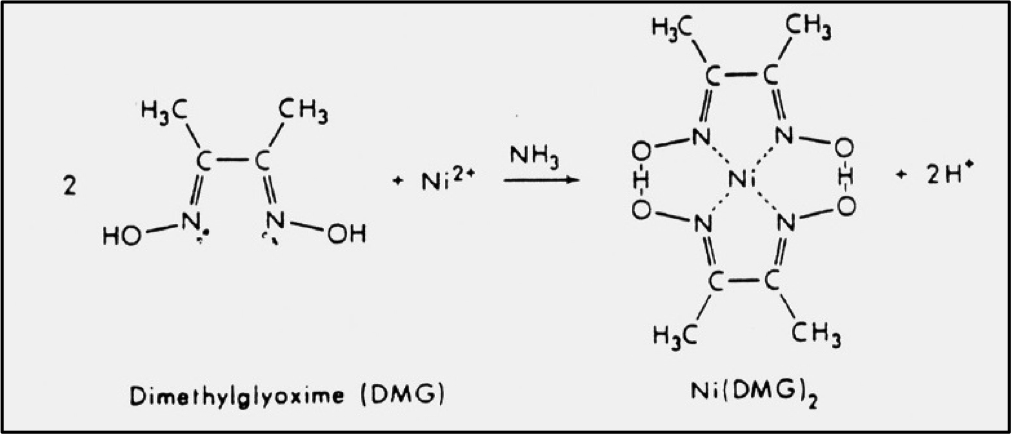
This site explains how to find molar mass. 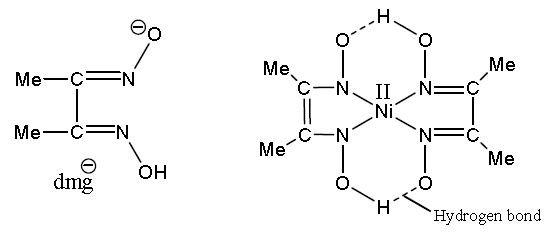
The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. Immediately after adding the acids, close the vessels, using the lid torque tool to ensure that the lids are closed appropriately. Add 8 mL of concentrated nitric acid and 3.5 mL hydrochloric acid (in the hood). Accurately weigh three 1.0 to 1.2-g samples of the dried unknown into clean, dry Teflon PFA microwave vessels (Figure 1).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
